FDA is launching real-time monitoring of drug trials, with AstraZeneca and Amgen as first test cases
The agency says 45% of drug development time is currently "dead time" — real-time data access could cut months or years from the process



















































































































.jpg?branch=production&format=jpg&width=1024)

























































































































































































































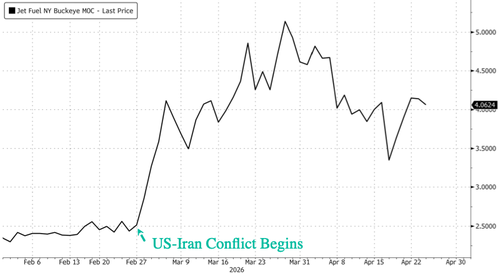











































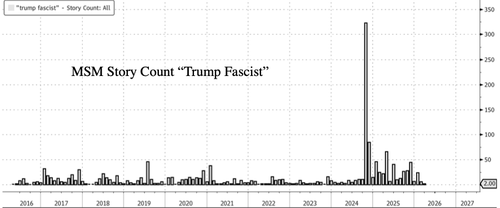






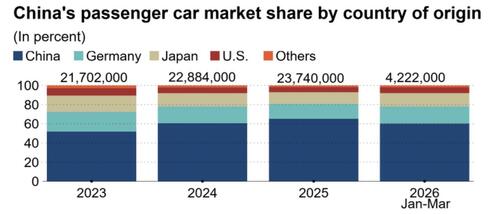



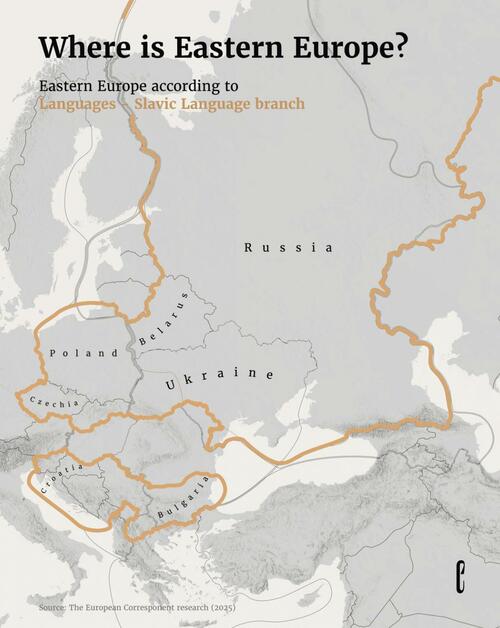






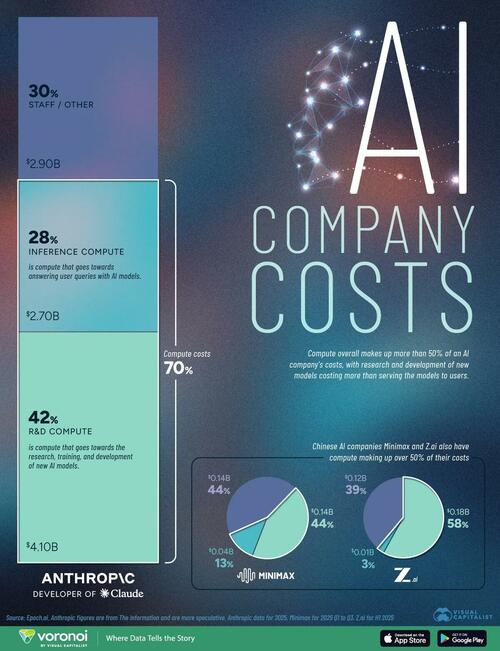

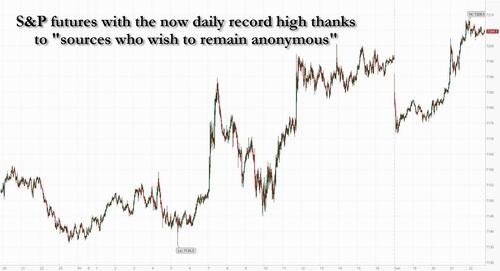







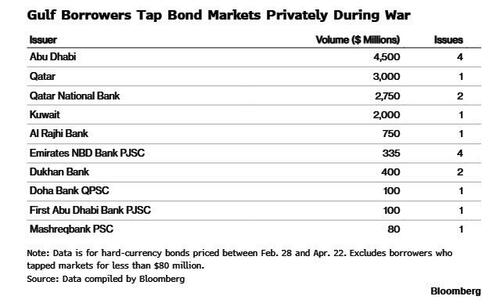








































































































































































































































.png?branch=production&format=jpg&width=1024)
























































































































































































































































































































































































































































































































































































































































.jpg?branch=production&format=jpg&width=1024)